- Home
- About CTI
- Our Services
- Investor Relations
- CTI Mall
- Resource Center
- Reports Validation
- Join Us
-
 Overview
Overview
Centre Testing International Group Co., Ltd. (CTI) is a market leader in testing, inspection, certification, calibration, audit, training & technical services; building trust between governments, enterprises, and consumers.
-
 Sustainability
SustainabilitySustainability is deeply rooted in CTI’s business model, by delivering science-based solutions and verification services, to increase transparency and traceability throughout the global value chain. CTI is a proponent of carbon neutrality and sustainable development.
-
 Our service
Our serviceCentre Testing International Co., Ltd. (CTI) is the pioneer and leader in the TIC Industry which provides one-stop solutions on testing, inspection, certification, calibration, audit, training & technical services.
-
By Industry
Our service capabilties cover the upstream and downstream of the supply chain including textile and apparel,toys,electronic appliances,medical health,food...andother industries.
-
 Environment
Environment
-
 Raw Material & Fuel Chemicals
Raw Material & Fuel Chemicals
-
 Textiles, Apparel, Footwear & Accessories
Textiles, Apparel, Footwear & Accessories
-
 Food & Agricultural Products
Food & Agricultural Products
-
 Cosmetics, Personal Care & Household Chemicals
Cosmetics, Personal Care & Household Chemicals
-
 Building Materials&Construction Engineering
Building Materials&Construction Engineering
-
 Electronic & Electrical Appliances
Electronic & Electrical Appliances
-
 Toys, Furniture & Home Decoration
Toys, Furniture & Home Decoration
-
 Industrial Equipment & Manufacturing
Industrial Equipment & Manufacturing
-
 Rail & Aviation
Rail & Aviation
-
 Automotive & Spare Parts
Automotive & Spare Parts
-
 Pharma and Medical Services
Pharma and Medical Services
-
 Maritime Vessel Compliance Testing
Maritime Vessel Compliance Testing
 By Industry
By IndustryOur service capabilties cover the upstream and downstream of the supply chain including textile and apparel,toys,electronic appliances,medical health,food...andother industries.
-
-
 Specialty
SpecialtyComprehensively guarantee quality and safety, promote compliance and innovation, demonstrate brand competitiveness, and achieve higher quality, healthier, safer, and greener sustainable development.
-
 Management
ManagementWe have established a clear governance structure in accordance with listing requirements and national regulations and policies to deal with internal and external challenges and achieve sustainable development.
-
 Information DisclosureWe are committed to establishing normal and effective two-way communication with shareholders and investors. We have established a complete information disclosure mechanism to convey information to shareholders in a timely manner.
Information DisclosureWe are committed to establishing normal and effective two-way communication with shareholders and investors. We have established a complete information disclosure mechanism to convey information to shareholders in a timely manner.
-
 Talents Policy
Talents PolicyEnsuring the basic rights and benefits of employees;
Providing professional skills training to promote employees’ growth;
Carrying out various kinds of activities to balance employees’ work and life.
-
 RecruitmentWelcome to join CTI family! We are providing a platform for you to show your talents and achieve your career aspiration.
RecruitmentWelcome to join CTI family! We are providing a platform for you to show your talents and achieve your career aspiration.
- Resource Center
- Application Forms
- Bulletin
- Training Center
- CTI Academy
- Reports Validation

QUALITY & VALUE
Biological products are a type of products used for prevention, treatment and diagnosis, such as antibody drugs, vaccines, gene and cell therapy drugs, etc., prepared from biological materials such as microorganisms, cells, tissues, and body fluids. Due to their different production methods from chemical drugs, biological products need to undergo strict biosafety testing at all stages, including raw materials, virus banks, cell banks, harvested fluids, and stock fluids. CTI Pharma and Medical Services· provides you with a variety of exogenous contamination factor inspections at all stages of biological product production.

Virus testing
Virus contamination inspection of biological products can be divided into extensive screening for unknown viruses and inspection for specific viruses. CTI will conduct internal and external virus contamination inspections based on customer needs in accordance with the relevant standards of the International Council for Harmonization of Technical Requirements for Registration of Pharmaceuticals for Human Use (ICH) Q5A, the General Chapter for Virus Safety Control of Biological Products of the Chinese Pharmacopoeia (ChP), the United States Pharmacopoeia (USP), and the European Pharmacopoeia (EP). Our testing items include:
- Virus titer determination
- Exogenous virus screening based on in vitro cell culture
- Specific virus inspection based on nucleic acid amplification technology (NAT)
Mycoplasma Testing
Mycoplasma is a common contaminating bacterium in cell culture, which is very easy to spread on a large scale. It not only affects cell metabolism, resulting in a decrease in the yield and quality of the final product, but is also related to many human diseases. Regulatory authorities in various countries have stipulated that mycoplasma contamination detection should be carried out in the production process of biological products. CTI provides three mycoplasma detection strategies based on the pharmacopoeia method (ChP3301, EP 2.6.7, USP 63): culture method, indicator cell culture method (DNA staining method), and nucleic acid amplification method (NAT).
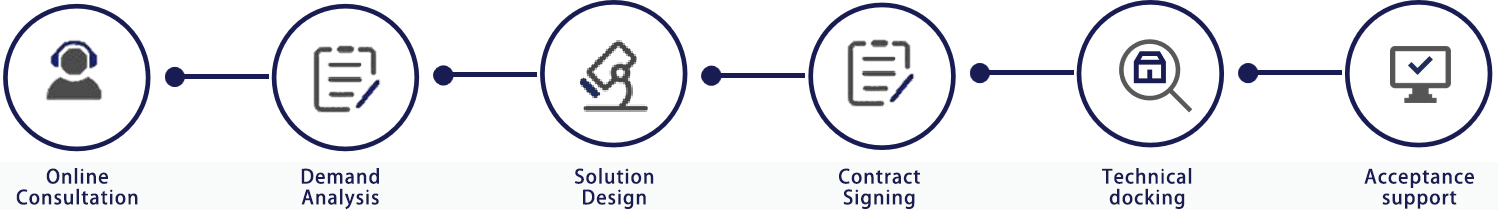
Service process
- About CTI
- Our Services
- Investor Relations
- CTI Mall
-
Resource Center
- Application Forms
- Bulletin
- Training Center
- CTI Academy
- Reports Validation
-
Join Us
- Talents Policy
- Recruitment



























 粤公网安备 44030602000441号
粤公网安备 44030602000441号 