- Home
- About CTI
- Our Services
- Investor Relations
- CTI Mall
- Resource Center
- Reports Validation
- Join Us
-
 Overview
Overview
Centre Testing International Group Co., Ltd. (CTI) is a market leader in testing, inspection, certification, calibration, audit, training & technical services; building trust between governments, enterprises, and consumers.
-
 Sustainability
SustainabilitySustainability is deeply rooted in CTI’s business model, by delivering science-based solutions and verification services, to increase transparency and traceability throughout the global value chain. CTI is a proponent of carbon neutrality and sustainable development.
-
 Our service
Our serviceCentre Testing International Co., Ltd. (CTI) is the pioneer and leader in the TIC Industry which provides one-stop solutions on testing, inspection, certification, calibration, audit, training & technical services.
-
By Industry
Our service capabilties cover the upstream and downstream of the supply chain including textile and apparel,toys,electronic appliances,medical health,food...andother industries.
-
 Environment
Environment
-
 Raw Material & Fuel Chemicals
Raw Material & Fuel Chemicals
-
 Textiles, Apparel, Footwear & Accessories
Textiles, Apparel, Footwear & Accessories
-
 Food & Agricultural Products
Food & Agricultural Products
-
 Cosmetics, Personal Care & Household Chemicals
Cosmetics, Personal Care & Household Chemicals
-
 Building Materials&Construction Engineering
Building Materials&Construction Engineering
-
 Electronic & Electrical Appliances
Electronic & Electrical Appliances
-
 Toys, Furniture & Home Decoration
Toys, Furniture & Home Decoration
-
 Industrial Equipment & Manufacturing
Industrial Equipment & Manufacturing
-
 Rail & Aviation
Rail & Aviation
-
 Automotive & Spare Parts
Automotive & Spare Parts
-
 Pharma and Medical Services
Pharma and Medical Services
-
 Maritime Vessel Compliance Testing
Maritime Vessel Compliance Testing
 By Industry
By IndustryOur service capabilties cover the upstream and downstream of the supply chain including textile and apparel,toys,electronic appliances,medical health,food...andother industries.
-
-
 Specialty
SpecialtyComprehensively guarantee quality and safety, promote compliance and innovation, demonstrate brand competitiveness, and achieve higher quality, healthier, safer, and greener sustainable development.
-
 Management
ManagementWe have established a clear governance structure in accordance with listing requirements and national regulations and policies to deal with internal and external challenges and achieve sustainable development.
-
 Information DisclosureWe are committed to establishing normal and effective two-way communication with shareholders and investors. We have established a complete information disclosure mechanism to convey information to shareholders in a timely manner.
Information DisclosureWe are committed to establishing normal and effective two-way communication with shareholders and investors. We have established a complete information disclosure mechanism to convey information to shareholders in a timely manner.
-
 Talents Policy
Talents PolicyEnsuring the basic rights and benefits of employees;
Providing professional skills training to promote employees’ growth;
Carrying out various kinds of activities to balance employees’ work and life.
-
 RecruitmentWelcome to join CTI family! We are providing a platform for you to show your talents and achieve your career aspiration.
RecruitmentWelcome to join CTI family! We are providing a platform for you to show your talents and achieve your career aspiration.
- Resource Center
- Application Forms
- Bulletin
- Training Center
- CTI Academy
- Reports Validation

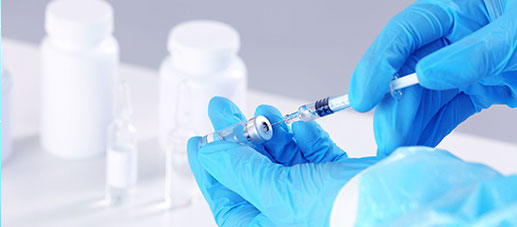
QUALITY & VALUE
CTI Pharma and Medical Services · CMC Services provides you with technology transfer services for Olopatadine hydrochloride eye drops. It has a team of senior experts and an advanced technology platform. It is committed to providing technical support for the entire process from API to formulation development, helping customers to efficiently complete drug research and development and production transformation, ensure product quality and compliance, and accelerate the process of drug launch.
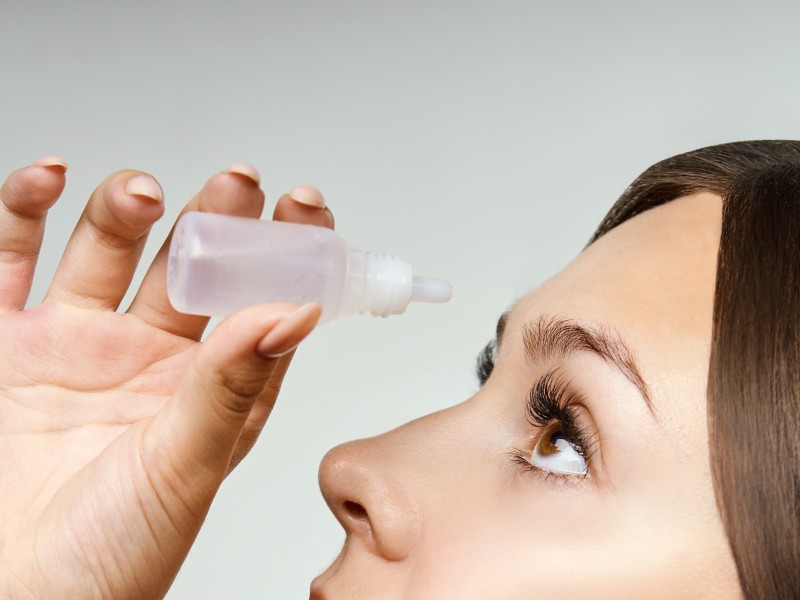
Service background
Allergic conjunctivitis is caused by hypersensitivity of the conjunctiva to allergens, mainly manifested as type I and type IV hypersensitivity reactions. At present, our country lacks large-sample epidemiological data. In the United States, 40% of people suffer from this disease, but only 10% seek medical treatment. In Japan, 15% to 20% of people have a history of allergic conjunctivitis. More than 90% of patients in the United States suffer from seasonal allergic conjunctivitis, while in China, perennial and seasonal allergic conjunctivitis account for 74%.
In addition, the market size of ophthalmic drugs in China has grown from $1.8 billion USD in 2015 to $2.6 billion USD in 2019, with a compound annual growth rate of 9.3%. The size of China's ophthalmic drug market is only a small fraction of that of the United States (about 18%), indicating that there is a huge gap in China's ophthalmic drug medical needs.
Technology transfer of olopatadine hydrochloride eye drops
1. Product overview
Generic name: olopatadine
Indications: allergic conjunctivitis
Dosage form: multiple doses 0.2% specification
Usage and dosage: once a day
Marketing status: Launched in China in 2022
Patent status: no patent restrictions
Registration category: new 4 categories
2. Therapeutic status and future trends
Antihistamine and mast cell stabilizer dual action drug: As the first choice of basic drugs, this type of drug can stabilize the mast cell membrane and antagonize histamine at the same time, and has a good control effect on both acute and intermittent inflammatory reactions, and is well tolerated.
Glucocorticoid drugs: Topical use on eyes can effectively inhibit immune cell activation and inflammatory mediator release, and are suitable for severe or recurrent allergic conjunctivitis, but the use time should not be too long, and attention should be paid to monitoring the risk of complications.
Immunosuppressants: Such as Cyclosporine A and Tacrolimus, topical use on eyes can inhibit a variety of inflammatory mediators, and are suitable for severe allergic conjunctivitis, especially for patients who are intolerant to glucocorticoids. There is a lack of long-term safety data in the clinic, and the condition needs to be closely observed and the medication should be adjusted when used.
3. The popularity and progress of R&D
11 companies have approved the 0.1% specification; 3 companies have been approved for the 0.2% specification; Production verification has been completed and the stability is 3 months.
Our advantages
Use once a day, more convenient than 0.1% Olopatadine eye drops; One of the best drugs for allergic conjunctivitis, the market is growing rapidly; Currently, only the original research is on the market, and the generic has not yet been approved for domestic listing;
Price floor
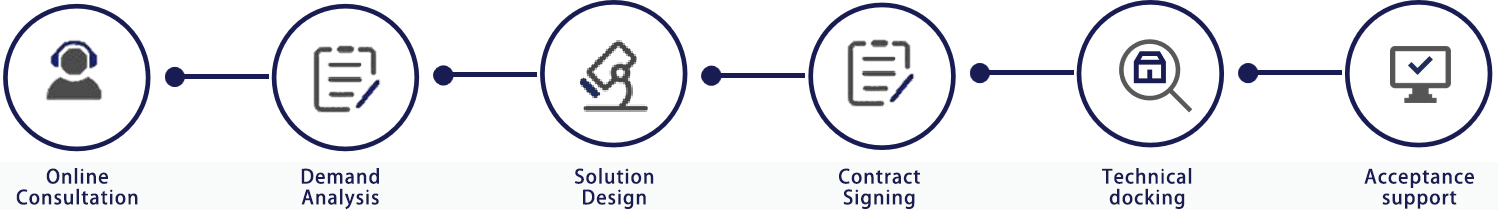
Service process
- About CTI
- Our Services
- Investor Relations
- CTI Mall
-
Resource Center
- Application Forms
- Bulletin
- Training Center
- CTI Academy
- Reports Validation
-
Join Us
- Talents Policy
- Recruitment




























 粤公网安备 44030602000441号
粤公网安备 44030602000441号 