- Home
- About CTI
- Our Services
- Investor Relations
- CTI Mall
- Resource Center
- Reports Validation
- Join Us
-
 Overview
Overview
Centre Testing International Group Co., Ltd. (CTI) is a market leader in testing, inspection, certification, calibration, audit, training & technical services; building trust between governments, enterprises, and consumers.
-
 Sustainability
SustainabilitySustainability is deeply rooted in CTI’s business model, by delivering science-based solutions and verification services, to increase transparency and traceability throughout the global value chain. CTI is a proponent of carbon neutrality and sustainable development.
-
 Our service
Our serviceCentre Testing International Co., Ltd. (CTI) is the pioneer and leader in the TIC Industry which provides one-stop solutions on testing, inspection, certification, calibration, audit, training & technical services.
-
By Industry
Our service capabilties cover the upstream and downstream of the supply chain including textile and apparel,toys,electronic appliances,medical health,food...andother industries.
-
 Environment
Environment
-
 Raw Material & Fuel Chemicals
Raw Material & Fuel Chemicals
-
 Textiles, Apparel, Footwear & Accessories
Textiles, Apparel, Footwear & Accessories
-
 Food & Agricultural Products
Food & Agricultural Products
-
 Cosmetics, Personal Care & Household Chemicals
Cosmetics, Personal Care & Household Chemicals
-
 Building Materials&Construction Engineering
Building Materials&Construction Engineering
-
 Electronic & Electrical Appliances
Electronic & Electrical Appliances
-
 Toys, Furniture & Home Decoration
Toys, Furniture & Home Decoration
-
 Industrial Equipment & Manufacturing
Industrial Equipment & Manufacturing
-
 Rail & Aviation
Rail & Aviation
-
 Automotive & Spare Parts
Automotive & Spare Parts
-
 Pharma and Medical Services
Pharma and Medical Services
-
 Maritime Vessel Compliance Testing
Maritime Vessel Compliance Testing
 By Industry
By IndustryOur service capabilties cover the upstream and downstream of the supply chain including textile and apparel,toys,electronic appliances,medical health,food...andother industries.
-
-
 Specialty
SpecialtyComprehensively guarantee quality and safety, promote compliance and innovation, demonstrate brand competitiveness, and achieve higher quality, healthier, safer, and greener sustainable development.
-
 Management
ManagementWe have established a clear governance structure in accordance with listing requirements and national regulations and policies to deal with internal and external challenges and achieve sustainable development.
-
 Information DisclosureWe are committed to establishing normal and effective two-way communication with shareholders and investors. We have established a complete information disclosure mechanism to convey information to shareholders in a timely manner.
Information DisclosureWe are committed to establishing normal and effective two-way communication with shareholders and investors. We have established a complete information disclosure mechanism to convey information to shareholders in a timely manner.
-
 Talents Policy
Talents PolicyEnsuring the basic rights and benefits of employees;
Providing professional skills training to promote employees’ growth;
Carrying out various kinds of activities to balance employees’ work and life.
-
 RecruitmentWelcome to join CTI family! We are providing a platform for you to show your talents and achieve your career aspiration.
RecruitmentWelcome to join CTI family! We are providing a platform for you to show your talents and achieve your career aspiration.
- Resource Center
- Application Forms
- Bulletin
- Training Center
- CTI Academy
- Reports Validation

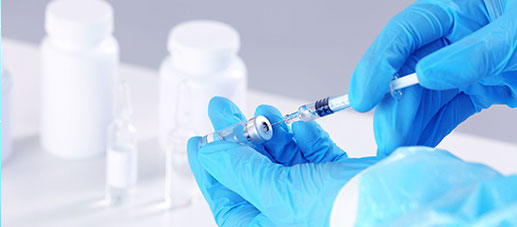
QUALITY & VALUE
CTI Pharma and Medical Services ·Drug Testing Services provides you with professional disinfectant efficacy verification services. It has an advanced pharmaceutical testing platform and an experienced experimental team. It is committed to ensuring the quality and safety of drugs; promoting the development and technological innovation of the pharmaceutical and health fields.

Service Background
Since cleaning and disinfection have a direct impact on the production of drugs/medical devices, cleaning and disinfection activities have always been the focus of government regulatory agencies. In order to confirm the applicability and disinfection efficacy of disinfectants or sporicides, CTI Pharma and Medical Services Drug Testing Services can provide you with professional disinfectant efficacy verification services to ensure that the drug/medical device production can be maintained at an acceptable cleanliness level.
Disinfectant efficacy verification
CTI Pharma and Medical Services ·Drug Testing Services provides you with professional disinfectant efficacy verification services. It has an advanced pharmaceutical testing platform and an experienced experimental team. It is committed to ensuring the quality and safety of drugs; promoting the development and technological innovation of the pharmaceutical and health fields.
1. Neutralizer verification
Neutralizer verification is to confirm that the selected neutralizer has a good neutralizing effect on disinfectants and sporicides without affecting the growth of microorganisms. Neutralizer verification must be carried out before the suspension germicidal test and the carrier germicidal test to prove the ability of the neutralizer to neutralize disinfectants and sporicides.
2. Suspension sterilization test
The suspension germicidal test can be used to quickly screen different disinfectants and sporicides and determine their disinfection effectiveness. That is, a certain concentration of disinfectant is mixed with a microbial suspension, and the survival of microorganisms is observed within a specified time to evaluate the sterilization effect of the disinfectant.
When determining the sterilization and disinfection effect of clean room surfaces, the suspension germicidal test cannot replace the carrier germicidal test.
3. Carrier germicidal test
Carrier germicidal test is used to detect the level of microorganism reduction of disinfectants and sporicides, and simulation tests are required on different types of surface materials. Among them, the materials used in the test should be selected from the representative ones in the actual scenarios of the manufacturer, and the risk of microorganisms attached to the surface of the material to the final product should be considered at the same time.
4. Disinfectant validity period verification
The unopened storage period of disinfectants shall not exceed the period specified in the written procedure. Generally, the validity period of disinfectants in sealed containers or initial packaging is closely related to the validity period provided by the manufacturer. However, once the initial packaging container (pre-formulated or concentrated) is unsealed, the validity period provided by the manufacturer will no longer apply to the concentration and sterility of the active substance in the disinfectant.
Therefore, the validity period verification of disinfectants usually focuses on the expiration date after the bottle is opened, that is, confirming the ability of the solution to destroy microorganisms within a certain period of time, and the time for which the appropriate bioburden level of the container itself and the solution is maintained in a controlled area.
CTI Pharma and Medical Services will evaluate and verify the shelf life of the opened bottle from two dimensions:
● Ensure the compliance of its disinfection effectiveness through sterilization tests;
● Determine the bioburden level through bioburden tests or sterility tests.
5. Customized disinfectant efficacy verification protocol and report
●Recommend disinfectant or sporicide type and cleaning/disinfection method based on the potential microbial contamination, equipment and facility materials and layout of the customer's actual site;
● Recommend test strains, test carriers and contact time based on the customer's disinfectant usage scenario;
● Develop a verification protocol and report that ultimately meets the regulatory requirements of various countries.
Applicable objects
The disinfectant efficacy verification service is applicable to the following objects:
● Various commonly used disinfectants and sporicides: Chlorine-containing disinfectants, Peroxide disinfectants, Aldehyde disinfectants, Alcohol disinfectants, Bromine-containing disinfectants, phenol disinfectants, etc.,
● Various surface materials: Stainless steel, Glass, PVC, Rubber, Color coated steel, Epoxy resin, etc.
● Various microorganisms: Staphylococcus aureus, Escherichia coli, Pseudomonas aeruginosa, Aspergillus niger, Candida albicans, Bacillus subtilis, environmental bacteria
Our advantages
CTI Pharma and Medical Services Drug testing services are committed to providing disinfectant efficacy verification services that meet the GMP requirements of various countries. With a professional team, advanced equipment and strict quality control, we ensure that we provide customers with high-quality and reliable verification services. We look forward to working with you to ensure the safety and effectiveness of pharmaceutical and medical device products.
1. Professional technical work team: The team has deep professional knowledge and rich practical experience, that can accurately perform disinfectant effect verification.
2. Strict quality control: Implement the cGMP system and implement strict quality control processes to ensure the authority of the test results.
3. Rich testing experience: The laboratory has rich experience in disinfectant testing, including but not limited to: 75% Alcohol, 70% Isopropyl alcohol (IPA), 6% Hydrogen peroxide, Acid phenol, Alkaline phenol, Sporicide, Compound Peracetic Acid, Bromo-geraminum, Quaternary ammonium salts, etc.
4. Customized services: expedited testing services, rigorous verification protocols and reports.
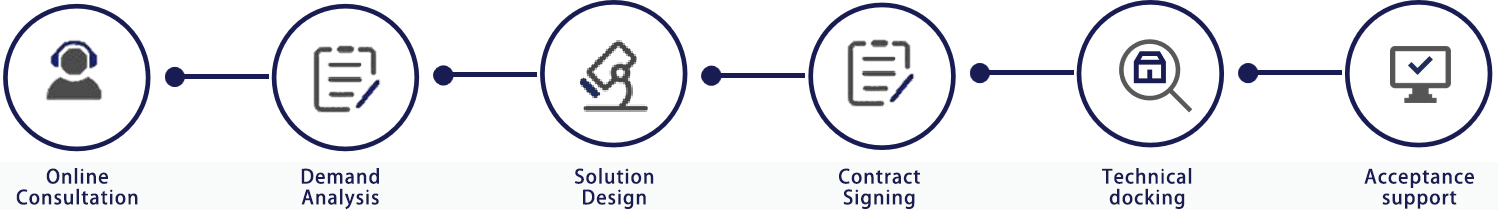
Service process
- About CTI
- Our Services
- Investor Relations
- CTI Mall
-
Resource Center
- Application Forms
- Bulletin
- Training Center
- CTI Academy
- Reports Validation
-
Join Us
- Talents Policy
- Recruitment



























 粤公网安备 44030602000441号
粤公网安备 44030602000441号 