- Home
- About CTI
- Our Services
- Investor Relations
- CTI Mall
- Resource Center
- Reports Validation
- Join Us
-
 Overview
Overview
Centre Testing International Group Co., Ltd. (CTI) is a market leader in testing, inspection, certification, calibration, audit, training & technical services; building trust between governments, enterprises, and consumers.
-
 Sustainability
SustainabilitySustainability is deeply rooted in CTI’s business model, by delivering science-based solutions and verification services, to increase transparency and traceability throughout the global value chain. CTI is a proponent of carbon neutrality and sustainable development.
-
 Our service
Our serviceCentre Testing International Co., Ltd. (CTI) is the pioneer and leader in the TIC Industry which provides one-stop solutions on testing, inspection, certification, calibration, audit, training & technical services.
-
By Industry
Our service capabilties cover the upstream and downstream of the supply chain including textile and apparel,toys,electronic appliances,medical health,food...andother industries.
-
 Environment
Environment
-
 Raw Material & Fuel Chemicals
Raw Material & Fuel Chemicals
-
 Textiles, Apparel, Footwear & Accessories
Textiles, Apparel, Footwear & Accessories
-
 Food & Agricultural Products
Food & Agricultural Products
-
 Cosmetics, Personal Care & Household Chemicals
Cosmetics, Personal Care & Household Chemicals
-
 Building Materials&Construction Engineering
Building Materials&Construction Engineering
-
 Electronic & Electrical Appliances
Electronic & Electrical Appliances
-
 Toys, Furniture & Home Decoration
Toys, Furniture & Home Decoration
-
 Industrial Equipment & Manufacturing
Industrial Equipment & Manufacturing
-
 Rail & Aviation
Rail & Aviation
-
 Automotive & Spare Parts
Automotive & Spare Parts
-
 Pharma and Medical Services
Pharma and Medical Services
-
 Maritime Vessel Compliance Testing
Maritime Vessel Compliance Testing
 By Industry
By IndustryOur service capabilties cover the upstream and downstream of the supply chain including textile and apparel,toys,electronic appliances,medical health,food...andother industries.
-
-
 Specialty
SpecialtyComprehensively guarantee quality and safety, promote compliance and innovation, demonstrate brand competitiveness, and achieve higher quality, healthier, safer, and greener sustainable development.
-
 Management
ManagementWe have established a clear governance structure in accordance with listing requirements and national regulations and policies to deal with internal and external challenges and achieve sustainable development.
-
 Information DisclosureWe are committed to establishing normal and effective two-way communication with shareholders and investors. We have established a complete information disclosure mechanism to convey information to shareholders in a timely manner.
Information DisclosureWe are committed to establishing normal and effective two-way communication with shareholders and investors. We have established a complete information disclosure mechanism to convey information to shareholders in a timely manner.
-
 Talents Policy
Talents PolicyEnsuring the basic rights and benefits of employees;
Providing professional skills training to promote employees’ growth;
Carrying out various kinds of activities to balance employees’ work and life.
-
 RecruitmentWelcome to join CTI family! We are providing a platform for you to show your talents and achieve your career aspiration.
RecruitmentWelcome to join CTI family! We are providing a platform for you to show your talents and achieve your career aspiration.
- Resource Center
- Application Forms
- Bulletin
- Training Center
- CTI Academy
- Reports Validation

QUALITY & VALUE
CTI Pharma and Medical Services· CMC Service provides you with Tacrolimus sustained-release capsule technology transfer services. With a team of senior experts and an advanced technology platform. It is committed to providing technical support for the entire process from API to drug development, helping customers to efficiently complete formulation research and production transformation, ensure product quality and compliance, accelerate the drug launch process.

Service Background
As of July 2024, more than 20 domestic companies have imitated Cyclosporine (oral/injectable dosage forms). In 2023, the total market value of the market was 1.9 billion CNY, of which soft capsules accounted for 1.7 billion CNY.
In 2023, the total domestic market value of Tacrolimus dosage forms was 6.3 billion CNY, of which capsules accounted for 6.1 billion CNY and sustained-release capsules accounted for 160 million CNY. Tacrolimus sustained-release capsules have obvious advantages over ordinary capsules: improved renal function, stable blood drug concentration; positive impact on drug economy; effective control of blood sugar and triglyceride levels, reduced cardiovascular risks and gastrointestinal discomfort. Only need to be taken once a day.
Technology transfer of Tacrolimus sustained-release capsules
Generic name: Tacrolimus
Indications: Prevention of graft rejection after kidney transplantation. Prevention of graft rejection in the maintenance period after liver transplantation. Treatment of graft rejection after kidney or liver transplantation that cannot be controlled by other immunosuppressive drugs.
Dosage form: 0.5 mg, 1 mg
Usage and dosage: Once a day, take in the early morning. Take it on an empty stomach, at least 1 hour before or 2 ~3hours after food.
Reference formulation: Tacrolimus sustained-release capsules, Astellas, Advagraf®/Sinograf®, original research and import
Marketing status: 2007 (Europe), 2010 (China)
Patent status: Preparation formulation CN99806415 expired in 2019, no patent research and development risk
Registration category: New Class 4 application
National centralized procurement: None
Medical insurance status: Class B medical insurance
Treatment status and future trends
In order to improve the long-term survival rate of liver and kidney transplants, patients need to take immunosuppressants for a long time, including immune-inducing formulation (such as monoclonal or polyclonal antibodies) used to reduce acute rejection reactions in the early stage of transplantation, and maintenance treatment drugs for long-term prevention of rejection reactions (such as calcineurin inhibitors Tacrolimus, Cyclosporine, etc.). As the first-line anti-rejection drug, tacrolimus is the first choice due to its small dosage, strong efficacy and few adverse reactions. Most solid organ transplant patients use tacrolimus maintenance treatment.
The popularity and progress of R&D
As of July 2024, there are 18 API companies and 2 pharmaceutical formulation companies (Hangzhou Zhongmei Huadong Pharmaceutical Co., Ltd. and Chengdu Suncadia Medicine Co.,Ltd.) have been registered or passed the review. Among them, 5 are in the process of registration, and another 5 have completed BE registration. Tacrolimus sustained-release capsules have attracted attention due to their high technical difficulty. CTI Pharma and Medical Services CMC Service already has a mature production process and provides secondary development services.
Our advantages
● International recognition: As a first-line anti-rejection drug, Tacrolimus has better efficacy and safety than Cyclosporine;
● Superior properties: The sustained-release capsule form improves safety and ease of use, and only needs to be taken once a day;
● Simplified process: For Category 4 generic drug applications, only BE studies are required;
● Technical barriers: As a high-difficulty generic drug in the industry, it has certain technical thresholds;
● Mature technology: CTI Pharma and Medical Services has perfect production technology and secondary development capabilities.
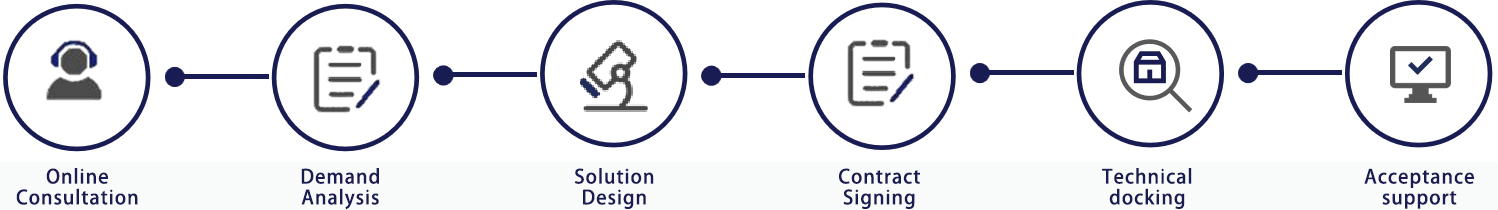
Service process
- About CTI
- Our Services
- Investor Relations
- CTI Mall
-
Resource Center
- Application Forms
- Bulletin
- Training Center
- CTI Academy
- Reports Validation
-
Join Us
- Talents Policy
- Recruitment




























 粤公网安备 44030602000441号
粤公网安备 44030602000441号 