- Home
- About CTI
- Our Services
- Investor Relations
- CTI Mall
- Resource Center
- Reports Validation
- Join Us
-
 Overview
Overview
Centre Testing International Group Co., Ltd. (CTI) is a market leader in testing, inspection, certification, calibration, audit, training & technical services; building trust between governments, enterprises, and consumers.
-
 Sustainability
SustainabilitySustainability is deeply rooted in CTI’s business model, by delivering science-based solutions and verification services, to increase transparency and traceability throughout the global value chain. CTI is a proponent of carbon neutrality and sustainable development.
-
 Our service
Our serviceCentre Testing International Co., Ltd. (CTI) is the pioneer and leader in the TIC Industry which provides one-stop solutions on testing, inspection, certification, calibration, audit, training & technical services.
-
By Industry
Our service capabilties cover the upstream and downstream of the supply chain including textile and apparel,toys,electronic appliances,medical health,food...andother industries.
-
 Environment
Environment
-
 Raw Material & Fuel Chemicals
Raw Material & Fuel Chemicals
-
 Textiles, Apparel, Footwear & Accessories
Textiles, Apparel, Footwear & Accessories
-
 Food & Agricultural Products
Food & Agricultural Products
-
 Cosmetics, Personal Care & Household Chemicals
Cosmetics, Personal Care & Household Chemicals
-
 Building Materials&Construction Engineering
Building Materials&Construction Engineering
-
 Electronic & Electrical Appliances
Electronic & Electrical Appliances
-
 Toys, Furniture & Home Decoration
Toys, Furniture & Home Decoration
-
 Industrial Equipment & Manufacturing
Industrial Equipment & Manufacturing
-
 Rail & Aviation
Rail & Aviation
-
 Automotive & Spare Parts
Automotive & Spare Parts
-
 Pharma and Medical Services
Pharma and Medical Services
-
 Maritime Vessel Compliance Testing
Maritime Vessel Compliance Testing
 By Industry
By IndustryOur service capabilties cover the upstream and downstream of the supply chain including textile and apparel,toys,electronic appliances,medical health,food...andother industries.
-
-
 Specialty
SpecialtyComprehensively guarantee quality and safety, promote compliance and innovation, demonstrate brand competitiveness, and achieve higher quality, healthier, safer, and greener sustainable development.
-
 Management
ManagementWe have established a clear governance structure in accordance with listing requirements and national regulations and policies to deal with internal and external challenges and achieve sustainable development.
-
 Information DisclosureWe are committed to establishing normal and effective two-way communication with shareholders and investors. We have established a complete information disclosure mechanism to convey information to shareholders in a timely manner.
Information DisclosureWe are committed to establishing normal and effective two-way communication with shareholders and investors. We have established a complete information disclosure mechanism to convey information to shareholders in a timely manner.
-
 Talents Policy
Talents PolicyEnsuring the basic rights and benefits of employees;
Providing professional skills training to promote employees’ growth;
Carrying out various kinds of activities to balance employees’ work and life.
-
 RecruitmentWelcome to join CTI family! We are providing a platform for you to show your talents and achieve your career aspiration.
RecruitmentWelcome to join CTI family! We are providing a platform for you to show your talents and achieve your career aspiration.
- Resource Center
- Application Forms
- Bulletin
- Training Center
- CTI Academy
- Reports Validation

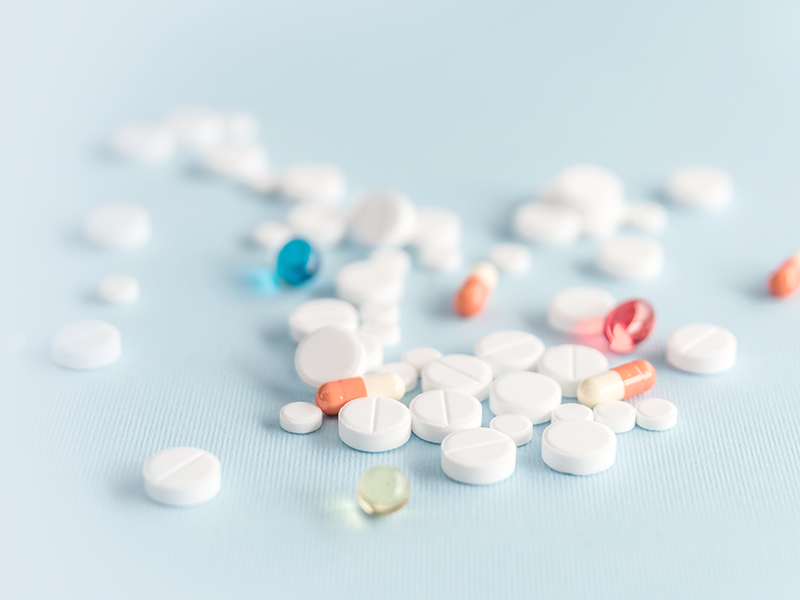
QUALITY & VALUE
API production processes include chemical synthesis, semi-synthesis, microbial fermentation and plant and animal extraction. Microbial fermentation is favored for its mild conditions and high yield. Control of impurities in fermentation and plant and animal extracts has become an indispensable part of the quality study of APIs.CTI can provide you with the residue analysis of DNA, protein and sugar in fermentation APIs (APIs).

Service Introduction
Bio-enzyme catalyzed reaction has the advantages of high specificity, mild conditions, green and environmental protection, etc., and has been more and more widely used in the production process of APIs. According to the type of reaction catalyzed by enzymes, the International Enzyme Commission (IE.C) classifies enzymes into 6 categories: transferases, hydrolases, cleavage enzymes, oxidoreductases, isomerases and ligases. The enzyme catalysts used in the preparation of AP| are process impurities and need to be controlled for residual amounts.
- Protein Residues
Protein content determination methods include Kjeldahl method, Folinol method (Lowry method), bis(acetonitrile) method, BCA method and Caulmers Brilliant Blue method (Bradford method), of which Caulmers Brilliant Blue method is favored because of the highest sensitivity and not easy to be interfered. This method can determine the amount of protein from 1 to 200 μg.
- DNA Residue
CTI adopts fluorescent staining method to detect the DNA residues of fermentation microorganisms in APIs.
- Sugar Residues
Whether it is starch, polysaccharides or oligosaccharides, the hydrolysis method can be used to convert sugars into monosaccharides, and the HPLC-CAD method can be used to detect the residues of sugars.
Service Background
Recently, CDE is concerned about the residues of enzymes, starch and DNA in the catalytic reaction during the new drug review of enzyme-catalyzed synthesized APIs. CTI Huatest has established the analytical methods for the residues of 3 components (DNA, protein and sugar).
Applicable Products
Applicable to enzyme-catalyzed, or fermentation-prepared APIs. Such as Vildagliptin, Norethindrone Hydrochloride, Atorvastatin Calcium, Rhodopsin, etc.
Service Content
Residue Detection and Methodology Validation of 3 Ingredients (DNA, Protein, Sugar) in APIs.
According to the Chinese Pharmacopoeia 2020 Edition IV “9101 Guiding Principles for Validation of Analytical Methods” for analytical methods and validation performance indicators, CTI performed methodological validation according to the requirements of impurity quantitative analytical methods. Parameters examined include specificity, linearity/range, accuracy, precision (repeatability/intermediate precision), limit of detection and limit of quantification, and solution stability.
The sensitivity of the method is such that it can fulfill most of the application scenarios.
|
Test Items |
Detection limit of the method |
Limit of quantification |
|
Protein Residue |
0.6µg/mL |
2µg/mL |
|
Sugar Residues |
1.7µg/mL |
5µg/mL |
|
DNA residues |
1.3ng/mL |
2.5ng/mL |
Specialty Services
For API soluble in water, only 2~3g of sample is required for single method detection, while 20~30g of sample is required for method development and validation.
- About CTI
- Our Services
- Investor Relations
- CTI Mall
-
Resource Center
- Application Forms
- Bulletin
- Training Center
- CTI Academy
- Reports Validation
-
Join Us
- Talents Policy
- Recruitment




























 粤公网安备 44030602000441号
粤公网安备 44030602000441号 