- Home
- About CTI
- Our Services
- Investor Relations
- CTI Mall
- Resource Center
- Reports Validation
- Join Us
-
 Overview
Overview
Centre Testing International Group Co., Ltd. (CTI) is a market leader in testing, inspection, certification, calibration, audit, training & technical services; building trust between governments, enterprises, and consumers.
-
 Sustainability
SustainabilitySustainability is deeply rooted in CTI’s business model, by delivering science-based solutions and verification services, to increase transparency and traceability throughout the global value chain. CTI is a proponent of carbon neutrality and sustainable development.
-
 Our service
Our serviceCentre Testing International Co., Ltd. (CTI) is the pioneer and leader in the TIC Industry which provides one-stop solutions on testing, inspection, certification, calibration, audit, training & technical services.
-
By Industry
Our service capabilties cover the upstream and downstream of the supply chain including textile and apparel,toys,electronic appliances,medical health,food...andother industries.
-
 Environment
Environment
-
 Raw Material & Fuel Chemicals
Raw Material & Fuel Chemicals
-
 Textiles, Apparel, Footwear & Accessories
Textiles, Apparel, Footwear & Accessories
-
 Food & Agricultural Products
Food & Agricultural Products
-
 Cosmetics, Personal Care & Household Chemicals
Cosmetics, Personal Care & Household Chemicals
-
 Building Materials&Construction Engineering
Building Materials&Construction Engineering
-
 Electronic & Electrical Appliances
Electronic & Electrical Appliances
-
 Toys, Furniture & Home Decoration
Toys, Furniture & Home Decoration
-
 Industrial Equipment & Manufacturing
Industrial Equipment & Manufacturing
-
 Rail & Aviation
Rail & Aviation
-
 Automotive & Spare Parts
Automotive & Spare Parts
-
 Pharma and Medical Services
Pharma and Medical Services
-
 Maritime Vessel Compliance Testing
Maritime Vessel Compliance Testing
 By Industry
By IndustryOur service capabilties cover the upstream and downstream of the supply chain including textile and apparel,toys,electronic appliances,medical health,food...andother industries.
-
-
 Specialty
SpecialtyComprehensively guarantee quality and safety, promote compliance and innovation, demonstrate brand competitiveness, and achieve higher quality, healthier, safer, and greener sustainable development.
-
 Management
ManagementWe have established a clear governance structure in accordance with listing requirements and national regulations and policies to deal with internal and external challenges and achieve sustainable development.
-
 Information DisclosureWe are committed to establishing normal and effective two-way communication with shareholders and investors. We have established a complete information disclosure mechanism to convey information to shareholders in a timely manner.
Information DisclosureWe are committed to establishing normal and effective two-way communication with shareholders and investors. We have established a complete information disclosure mechanism to convey information to shareholders in a timely manner.
-
 Talents Policy
Talents PolicyEnsuring the basic rights and benefits of employees;
Providing professional skills training to promote employees’ growth;
Carrying out various kinds of activities to balance employees’ work and life.
-
 RecruitmentWelcome to join CTI family! We are providing a platform for you to show your talents and achieve your career aspiration.
RecruitmentWelcome to join CTI family! We are providing a platform for you to show your talents and achieve your career aspiration.
- Resource Center
- Application Forms
- Bulletin
- Training Center
- CTI Academy
- Reports Validation

QUALITY & VALUE
CTI Pharma and Medical Services · Drug Testing Service provides you with MAH certification conversion services. It has an advanced pharmaceutical testing platform and an experienced experimental team. It is committed to ensuring the quality and safety of drugs; promoting the development and technological innovation of the pharmaceutical and health fields.

Service Background
In the pharmaceutical field, MAH certified conversion services help companies or individuals that have R&D and innovation capabilities but probably without production capabilities to successfully convert their R&D products into legally marketed products. Specifically, under the MAH (Marketing Authorization Holder) system, R&D units do not need to build their own production lines and can directly apply for drug marketing authorization certificates. MAH certified conversion service providers play the role of a bridge.
Drug MAH Certification Conversion Service
Service Purpose
We propose solutions around the entire process of how to make a drug legal, compliant and efficient from the laboratory to the market, helping R&D entities focus on their core R&D activities and reduce entry barriers and operating costs.
Service scope
1. Guide customers to establish MAH quality system and assist in applying for B license
2. Provide MAH holding service for enterprises without B license
3. Customize and develop target products according to customer needs and obtain approval
4. Implement suitable industrial bases
5. Provide quality management system construction services for drugs after lunched
6. Continuous product quality improvement services
Build a joint certification platform
1. Build a joint certification company
2. Provide a research and development industrialization platform
3. Provide a quality management system
4. Partners provide a sales platform
Service Targets
1. Innovative biotechnology companies
2. Small and medium-sized pharmaceutical companies
3. CROs
4. Drug holders or teams
Our advantages
1. Formulation Department
Lead by PhDs, with a master's degree with decades of work experience as research assistant, we are committed to the development and industrialization of oral solid formulation, lyophilized formulation, injections, ophthalmic formulation and complex injections. Multiple formulation project teams, fully equipped with: freeze drier, capping machine, dry granulation, wet granulation, process bed, coating machine, tablet press, capsule filling machine, high-pressure homogenizer and other formulation preparation equipment.
2. Raw Materials Department
Lead by senior PhDs, expert, team members have completed the development and industrialization of hundreds of difficult raw materials, and have obvious advantages in the fields of Carbapenems, -tinib, Gadolinium contrast agents, etc.
Equipped with 50 Test Batch fume hoods, hydrogenation reaction room and 100L Pilot Batch laboratory.
3. Quality Research Department
The domestic first-class quality research team has a high reputation in the fields of drug quality research, impurity research, and genotoxic impurity research. Team members have completed the development and verification of 500+ analytical methods and have rich experience and theoretical foundation.
Equipped with two-dimensional liquid chromatography from Agilent 1290 ultra-high performance liquid phase with 6530 time-of-flight mass spectrometer. QE from Thermo Fisher. There are more than 20 HPLCs, 2 GCs, ICP-MS, IC, TGA, DSC, UV, 4 automatic sampling dissolution instruments, conductivity, residual oxygen, and dissolved oxygen measuring instruments.
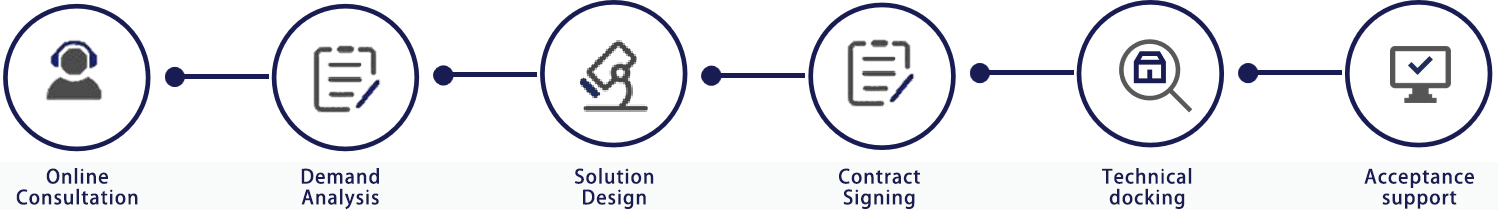
Service process
- About CTI
- Our Services
- Investor Relations
- CTI Mall
-
Resource Center
- Application Forms
- Bulletin
- Training Center
- CTI Academy
- Reports Validation
-
Join Us
- Talents Policy
- Recruitment



























 粤公网安备 44030602000441号
粤公网安备 44030602000441号 