- Home
- About CTI
- Our Services
- Investor Relations
- CTI Mall
- Resource Center
- Reports Validation
- Join Us
-
 Overview
Overview
Centre Testing International Group Co., Ltd. (CTI) is a market leader in testing, inspection, certification, calibration, audit, training & technical services; building trust between governments, enterprises, and consumers.
-
 Sustainability
SustainabilitySustainability is deeply rooted in CTI’s business model, by delivering science-based solutions and verification services, to increase transparency and traceability throughout the global value chain. CTI is a proponent of carbon neutrality and sustainable development.
-
 Our service
Our serviceCentre Testing International Co., Ltd. (CTI) is the pioneer and leader in the TIC Industry which provides one-stop solutions on testing, inspection, certification, calibration, audit, training & technical services.
-
By Industry
Our service capabilties cover the upstream and downstream of the supply chain including textile and apparel,toys,electronic appliances,medical health,food...andother industries.
-
 Environment
Environment
-
 Raw Material & Fuel Chemicals
Raw Material & Fuel Chemicals
-
 Textiles, Apparel, Footwear & Accessories
Textiles, Apparel, Footwear & Accessories
-
 Food & Agricultural Products
Food & Agricultural Products
-
 Cosmetics, Personal Care & Household Chemicals
Cosmetics, Personal Care & Household Chemicals
-
 Building Materials&Construction Engineering
Building Materials&Construction Engineering
-
 Electronic & Electrical Appliances
Electronic & Electrical Appliances
-
 Toys, Furniture & Home Decoration
Toys, Furniture & Home Decoration
-
 Industrial Equipment & Manufacturing
Industrial Equipment & Manufacturing
-
 Rail & Aviation
Rail & Aviation
-
 Automotive & Spare Parts
Automotive & Spare Parts
-
 Pharma and Medical Services
Pharma and Medical Services
-
 Maritime Vessel Compliance Testing
Maritime Vessel Compliance Testing
 By Industry
By IndustryOur service capabilties cover the upstream and downstream of the supply chain including textile and apparel,toys,electronic appliances,medical health,food...andother industries.
-
-
 Specialty
SpecialtyComprehensively guarantee quality and safety, promote compliance and innovation, demonstrate brand competitiveness, and achieve higher quality, healthier, safer, and greener sustainable development.
-
 Management
ManagementWe have established a clear governance structure in accordance with listing requirements and national regulations and policies to deal with internal and external challenges and achieve sustainable development.
-
 Information DisclosureWe are committed to establishing normal and effective two-way communication with shareholders and investors. We have established a complete information disclosure mechanism to convey information to shareholders in a timely manner.
Information DisclosureWe are committed to establishing normal and effective two-way communication with shareholders and investors. We have established a complete information disclosure mechanism to convey information to shareholders in a timely manner.
-
 Talents Policy
Talents PolicyEnsuring the basic rights and benefits of employees;
Providing professional skills training to promote employees’ growth;
Carrying out various kinds of activities to balance employees’ work and life.
-
 RecruitmentWelcome to join CTI family! We are providing a platform for you to show your talents and achieve your career aspiration.
RecruitmentWelcome to join CTI family! We are providing a platform for you to show your talents and achieve your career aspiration.
- Resource Center
- Application Forms
- Bulletin
- Training Center
- CTI Academy
- Reports Validation

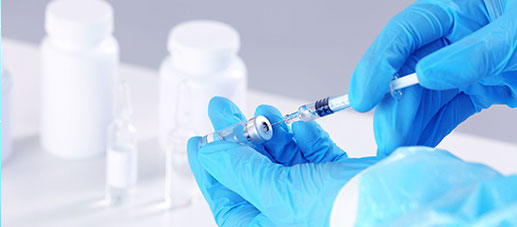
QUALITY & VALUE
CTI Pharma and Medical Services· Drug Testing Services provides you with professional bacteriophage testing services. It has an advanced pharmaceutical testing platform and an experienced experimental team. It is committed to ensuring the quality and safety of drugs; promoting the development and technological innovation of the pharmaceutical and health fields.
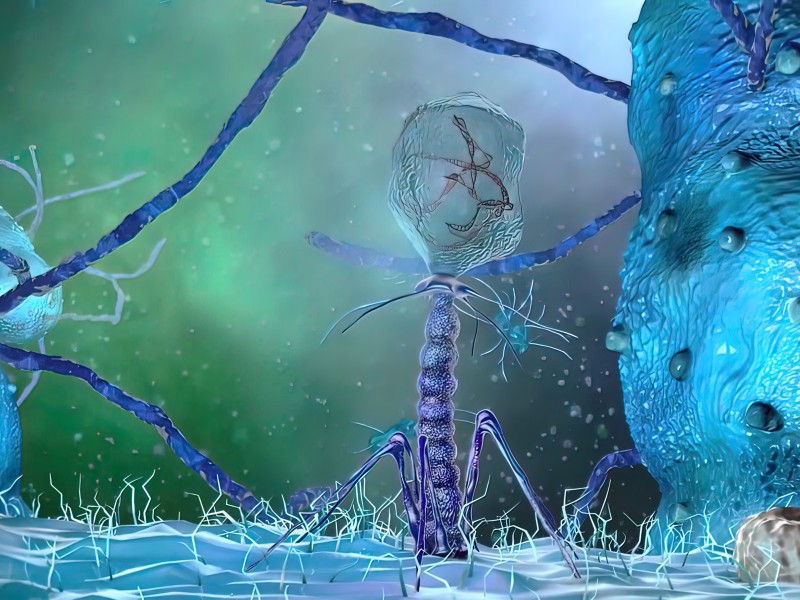
Service Background
In the process of drug production, bacteriophage control is an important link that cannot be ignored. Newborn calf serum is an important raw material in the production of biological products. Bacteriophage contamination will directly affect the quality of biological products, especially the production of live vaccines; and for E. coli seed banks, bacteriophage contamination may cause E. coli lysis, thereby affecting the stability and efficiency of the fermentation process, and then affecting the yield and quality of the product. Therefore, in order to ensure product quality, monitoring and control of bacteriophages is very necessary!
Bacteriophage Testing Service
CTI Pharma and Medical Services· Drug Testing Services provides you with professional bacteriophage testing services. It has an advanced pharmaceutical testing platform and an experienced experimental team. It is committed to ensuring the quality and safety of drugs; promoting the development and technological innovation of the pharmaceutical and health fields.
Test Method
Referring to ChP General Chapter 3604, two methods can be used to detect bacteriophages: double-layer plate-plaque method and proliferation method:
1.Plaque method: Lytic plaques are formed after a bacteriophage replicates and proliferates on a solid culture medium and lyses the host bacteria. By counting plaques, the number of phage units in a certain volume, that is, the number of phages, can be determined. In the double-layer plate-plaque method, first pour the bottom solid culture medium into the culture dish, and then pour the semi-solid culture medium containing host bacteria and a certain dilution of phages after solidification. After a period of culture, the number of plaques can be counted;
2.Proliferation method: First mix the sample with the host bacterial suspension to allow the phage in the sample to proliferate, then spot the sample liquid on the surface of the culture medium evenly covered with E. coli to observe the formation of plaques.
Service Content
1. Customized Validation Scheme: Develop a unique validation scheme for customer samples, issue a validation report, and confirm the reliability of the test method.
2. Detection of bacteriophage contamination in samples: The laboratory has completed internal validation of the double-layer plate-plaque method and proliferation method, and has established quality control procedures, and can perform phage testing on samples such as newborn calf serum or E. coli seed bank.
3.Customer communication and feedback: Establish open communication channels and encourage customers to provide feedback so that customer questions and concerns can be addressed in a timely manner. Provide customized testing services and solutions based on customer specific needs.
Service Advantages
1. Research methods: Use the latest research and industry standards to guide specific services.
2. Professional team: The team is composed of experienced microbiologists and quality control experts.
3. Customized services: Customized solutions can be provided according to the personalized needs of customers.
4. Continuous support: Provide continuous technical support and consulting services to meet customer needs in a timely manner.
Service process
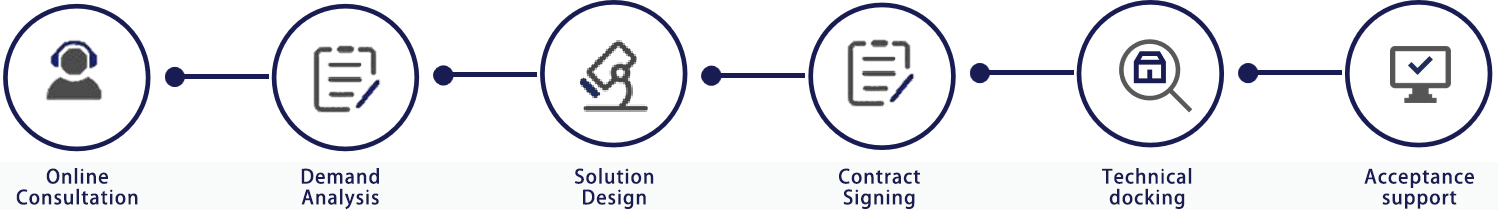
- About CTI
- Our Services
- Investor Relations
- CTI Mall
-
Resource Center
- Application Forms
- Bulletin
- Training Center
- CTI Academy
- Reports Validation
-
Join Us
- Talents Policy
- Recruitment




























 粤公网安备 44030602000441号
粤公网安备 44030602000441号 